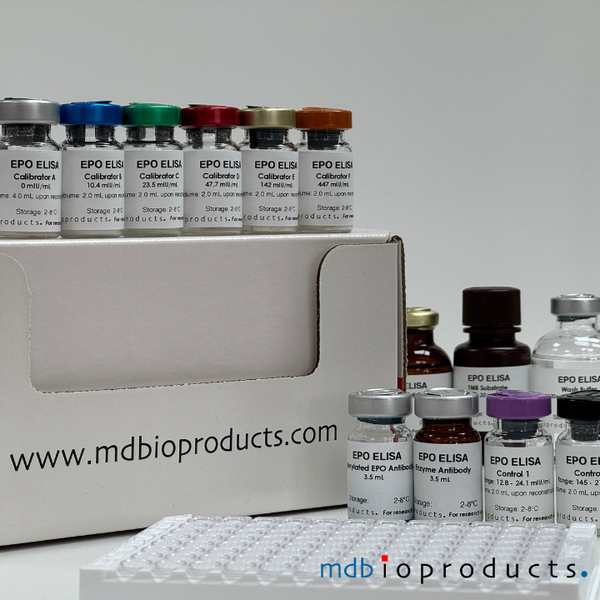
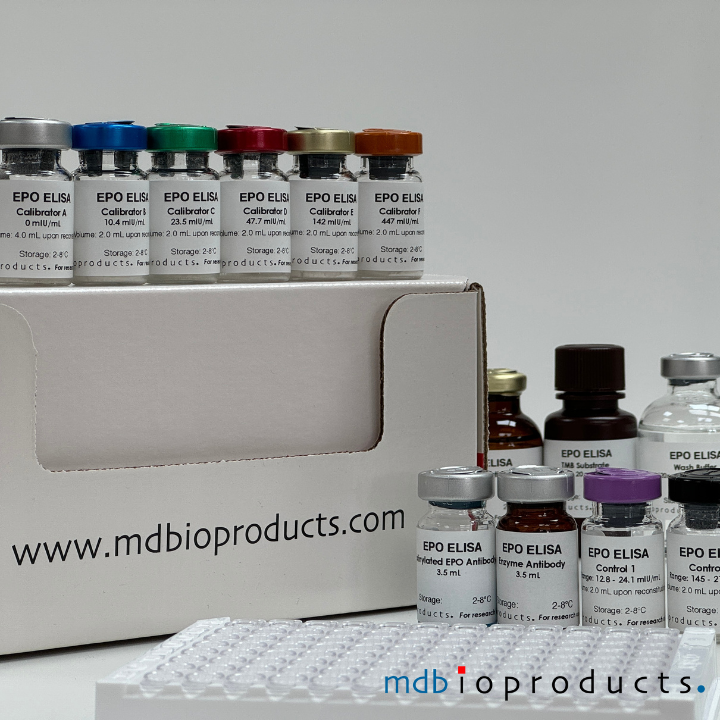
EPO ELISA
The EPO ELISA Kit is intended to measure Erythropoietin (EPO) levels in human serum in 2.5 hours. This assay is intended for research use, as an aid in the diagnosis of...
ELISA
M046013
The EPO ELISA Kit is intended to measure Erythropoietin (EPO) levels in human serum in 2.5 hours. This assay is intended for research use, as an aid in the diagnosis of anemias and polycythemias. With the advent of the administration of recombinant erythropoietin as a biologic therapy to increase red blood cell mass, an erythropoietin assay may be used also to aid in the prediction and monitoring of response to recombinant erythropoietin treatment in persons with anemias.
Erythropoietin (EPO) is a heavily glycosylated protein with a molecular weight of about 30,000 - 34,000 Daltons. Human EPO is a polypeptide consisting of 165 amino acids, containing one O-linked and three N-linked carbohydrate chains. The recombinant EPO is a good substitute for the native protein for use in an immunoassay. Serum EPO levels are dependent on the rate of production and the rate of clearance of the protein. Ninety percent of EPO is produced in the peritubular cells of the adult kidney in response to a decrease in tissue oxygenation. There is evidence indicating that the protein on these cells which detects oxygen saturation of the blood is a heme-containing moiety. As the pO2 of the plasma, a function of the hematocrit decreases, EPO concentration will increase. There are also observations suggesting that normally there is an inverse correlation between serum EPO levels and red blood cell mass.
Quantitation of serum erythropoietin concentration serves as a diagnostic adjunct in determining the cause of anemia or erythrocytosis. Aplastic anemia, hemolytic anemia and anemia due to iron deficiency all result in serum EPO elevation. Whereas, EPO levels in patients with secondary anemia due to renal failure and other disorders such as acquired immune deficiency syndrome (AIDS) are generally inappropriately low for the degree of anemia. This is mostly likely caused by an impaired ability of the diseased kidney to produce adequate quantities of EPO8. Low concentrations of EPO may give an early warning of kidney transplant rejection. EPO also can be used to monitor AIDS patients undergoing Zidovudine (AZT) therapy. An increased concentration of EPO verifies that anemia associated with AZT therapy is due to red cell hypoplasia or apliasia.
Polycythemia rubra vera, or primary erythrocytosis (an increase of red blood cell mass) results from unstimulated over production of erythrocytes. Hence, the increase in the hemoglobin causes decreased production of EPO, which results in subnormal levels of serum EPO. Secondary polycythemias, which are also characterized by an increase in the total red blood cell mass, occur as a physiological response to elevated levels of circulatory EPO caused by tissue hypoxia. The hypoxia may be due to such factors as pulmonary fibrosis, cardiovascular disease, prolonged exposure to high altitude, abnormal forms of hemoglobin or drug treatment. Some tumors produce EPO and, in these cases, EPO may be used as a tumor marker to monitor the effectiveness of treatment.
References/Citations:
Soliman NA, Mansour SW, Ammar MA, Ammar MAM. Pomegranate (Molasses, White Peel, Red Peel) effect in reducing Phenylhydrazine-induced Spleen injury and hemolytic anemia incidence. Research Square; 2023. DOI: 10.21203/rs.3.rs-2319307/v1.
Zhuang, X., Zhao, D., Yang, P., Jia, Y., Liang, R., Zhao, Q., ... & Li, J. (2018). 99mTc‐labeled rHuEpo for imaging of the erythropoietin receptor in tumors. Journal of Labelled Compounds and Radiopharmaceuticals, 61(2), 77-83.
Singh, A. K., Gupta, S., Barnes, A., Carlson, J. M., & Ayers, J. K. (2007). Red blood cell erythropoietin, not plasma erythropoietin, concentrations correlate with changes in hematological indices in horses receiving a single dose of recombinant human erythropoietin by subcutaneous injection. Journal of veterinary pharmacology and therapeutics, 30(2), 175-178.
AAV9-mediated erythropoietin gene delivery into the brain protects nigral dopaminergic neurons in a rat model of Parkinson's disease.
Xue YQ, et al. Gene Therapy. 2010; 17:83-94.Failure of erythropoietin to render jeopardized ischemic myocardium amenable to incremental salvage by early reperfusion.
French CJ, et al. Coron Artery Dis. 2009 Jun; 20(4):295-9.Erythropoietin to augment myocardial salvage induced by coronary thrombolysis in patients with ST segment elevation acute myocardial infarction.
Binbrek AS, et al. Am J. Cardiol. 2009 Oct; 104(8):1035-40.Analysis of recombinant human erythropoietin and darbepoietin in spiked plasma.
Singh AK, et al. Proteomics: Clinical Appl. 2007 Jun; 1(7):626-639
Product Insert (PDF) - Informational use only. Please refer to insert included with product.
Data/Specifications:
- Species: human
- Sample Type: serum
- Sample Size: 200 uL
- Standard Curve Range: 7.5 - 500 mU/mL
- Sensitivity: 1.2 mU/mL
- Assay Length: 2.5 hrs